Research Topics Institute of Inorganic Chemistry
Metal-based drugs are on the one hand indispensible in medicinal cancer therapy, but on the other hand their potential is far from being fully exploited or understood. Therefore, the main emphasis of our research is laid on the development of novel tumor-inhibiting metal complexes and organometallic compounds. As some of these classes of metal compounds contain biologically active ligand molecules (e.g., derivatives of naturally occurring compounds), chemical synthesis involves inorganic as well as organic components.
Synthesized compounds are thoroughly characterized by means of analytical and bioanalytical methods, including aspects such as stability in solution, interactions with biomolecules, and redox behavior. The capacity of inhibiting cancer cell growth is determined in cell culture tests, and structure–activity relationships within series of closely related compounds aid in directing further synthetic efforts. Furthermore, cell culture models serve for investigations into the modes of action of these compounds.
By cooperation with partners from the Medical University of Vienna within the cross-university research platform “Translational Cancer Therapy Research“, further mechanistic investigations as well as in vivo studies are performed. Thereby, a broad spectrum of preclinical anticancer drug research is spanned from chemical synthesis to biological activity studies.
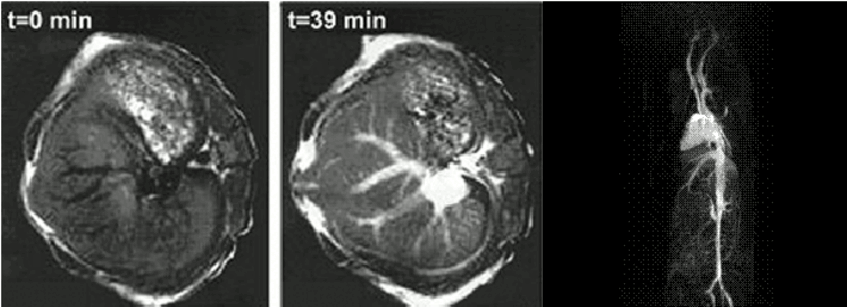
Another long-term project is devoted to the development of metal complexes for medical diagnostics, in particular gadolinium-based nanoparticular contrast agents for magnetic resonance imaging (MRI).
Compounds in clinical development
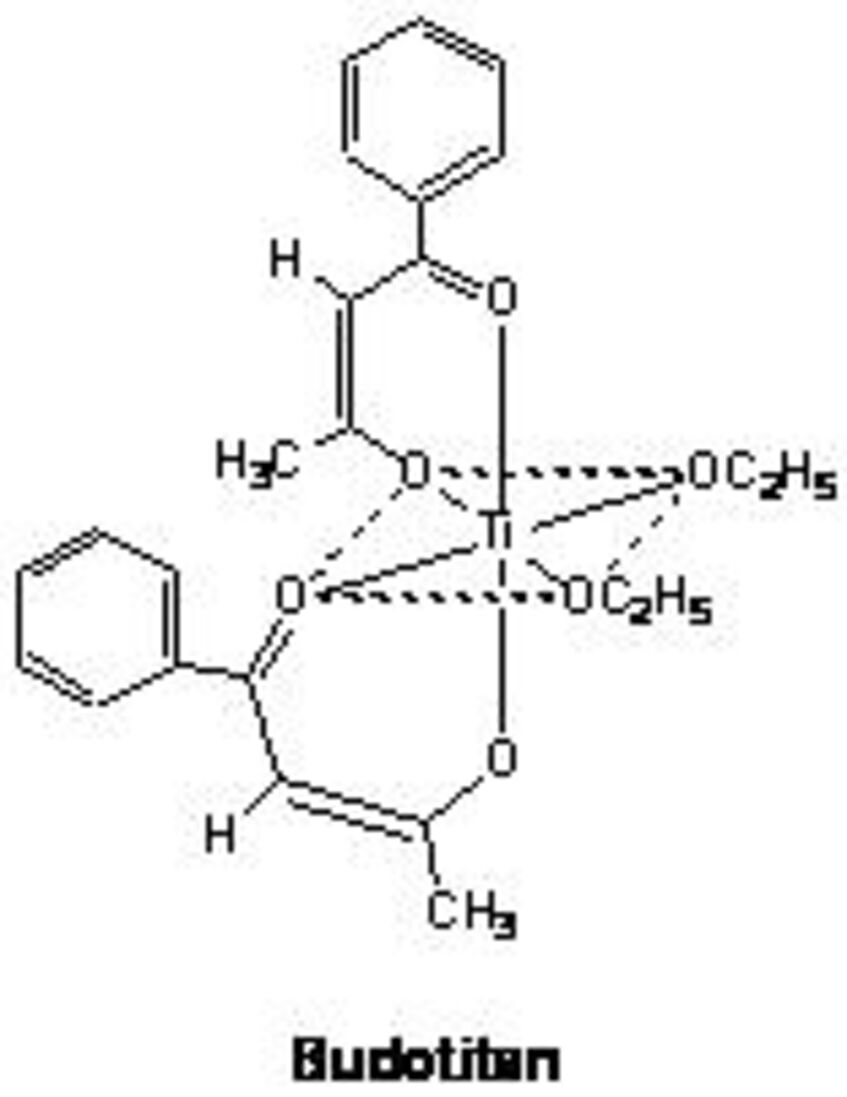
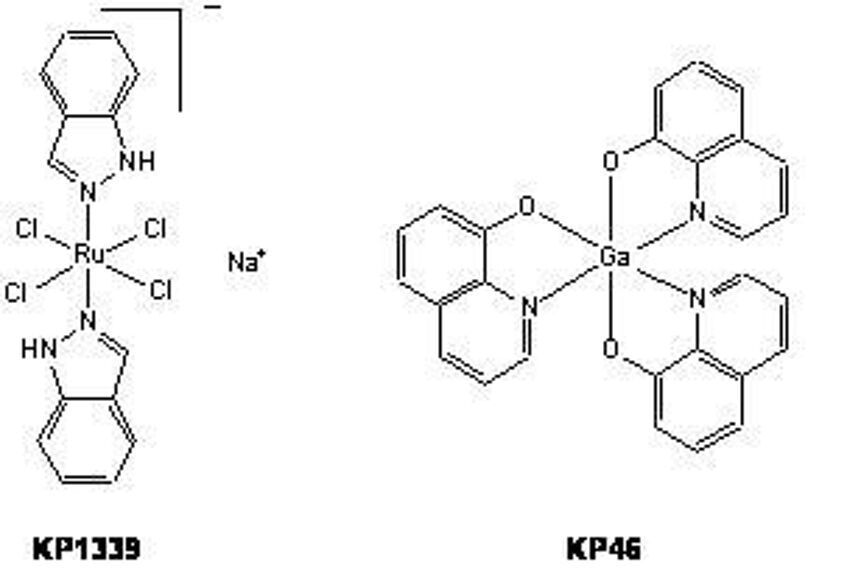
Budotitane (KP102), which had been developed in the past by this working group, was the second anticancer transition metal complex (after cisplatin) to be evaluated in clinical studies. Currently, two other compounds are in the stage of clinical development: the ruthenium complex sodium trans-[(tetrachloridobis(1H-indazole)ruthenate(III)] (KP1339) and the gallium complex tris(8-quinolinolato)gallium(III) (KP46).
Due to its affinity for serum proteins, the ruthenium compound KP1339 can accumulate in tumor tissue by the enhanced-permeability-and-retention effect (EPR effect) upon binding to albumin, and it can be transported into tumor cells via transferrin and the transferrin receptor (Trojan Horse hypothesis). Under the hypoxic conditions in solid tumors, the compound can be activated to more reactive ruthenium(II) species (activation-by-reduction hypothesis). Thus, treatment of tumors is possible with very low side effects. The mode of action involves generation of reactive oxygen species and interference with functions of the endoplasmic reticulum. A clinical phase I study in patients with advanced solid tumors has been finished with encouraging results (Thompson et al., ASCO Annual Meeting 2012). Most notably, patients with gastro-intestinal neuroendocrine tumors, for which currently no convincing medicinal therapies exist, benefited from the treatment.
The ruthenium compound acts like a Trojan horse and is accumulated in the tumour via transferrin and partly also via albumin, under the reductive and hypoxic conditions in tumours the compound is activated and treatment of tumours is possible with very small side effects. A clinical phase I study has been finished. All patients came in with progressive disease, five of six patients changed to stable disease.
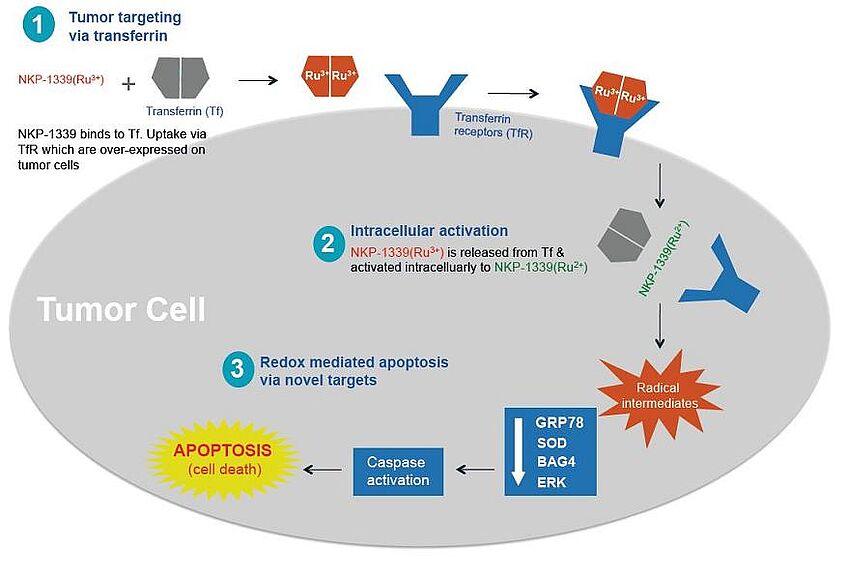
Mode of action of KP1339 (NKP-1339)
The gallium compound KP46 is an orally applied drug, which has also been evaluated in a clinical phase I trial. Remarkable activity was observed in patients with renal cell carcinoma (three of four patients showed therapeutic benefit). This compound exerts tumor-inhibiting effects probably by interference with iron metabolism.
Compounds in preclinical development (selected)
Oxaliplatin derivatives have been developed by adding small substituents to the parent complex with astonishing effects. A selected enantiomerically pure derivative (KP1537) proved to be superior to the parent drug in terms of efficacy, tolerability and therapeutic window in vivo. Side effects, in particular neurotoxicity, are milder, allowing the administration of higher doses.
Osteotropic platinum complexes have long been established by this working group, with KP735 as a prototypic compound. These complexes bind with high affinity to the hydroxylapatite of the bone by phosphonic acid groups and can be used to treat bone tumors and bone metastases.
A class of pH-sensitive platinum complexes have been developed, which are activated in the acidic milieu of solid tumors. Furthermore, platinum(IV) complexes have been synthesized, which are stable enough for oral administration and gastro-intestinal absorption and offer numerous possibilities for chemical modification.
Structures of selected platinum compounds:
Oxaliplatin derivative (KP1537), osteotropic complex (KP735), pH-sensitive complex (KP1421) and Pt(IV) complex.
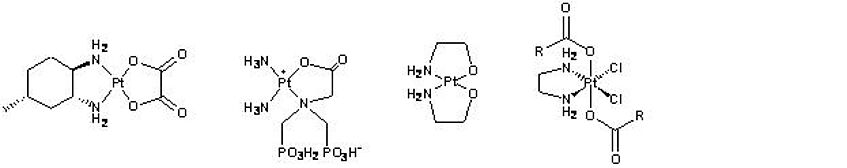
Gadolinium complexes are being developed to improve contrast media for magnetic resonance imaging. Nanoparticles have been synthesized and linked to lectins, which selectively accumulate at the endothelia of blood vessels and thereby enable the detection of atherosclerotic plaques as well as tumor blood vessels at an early stage.